Editor’s note
See the Hydroponic Nutrient Solutions Calculator (XLSX) for more information.
Nutrient solutions are a vital part of hydroponic crop care. Although production requirements can vary greatly between different hydroponic systems and different crops grown hydroponically, it is always important to maintain the quality of the nutrient solution. This publication outlines important considerations for understanding, formulating, and managing hydroponic nutrient solutions. This publication is divided into two parts. The first part describes how water quality parameters affect plant nutrient uptake. The second part outlines the management practices you need to follow to prepare nutrient solutions and promote nutrient availability.
The purpose of the nutrition program in controlled environment agriculture is to maintain the plant’s “comfort zone” around the roots. These are the main aspects of maintaining a plant’s comfort zone: • Provide all essential elements • Provide elements in optimum quantities for plant growth • Promote availability and absorption of nutrients
These three aspects often overlap. For example, you may have provided the optimum quantity of an essential element for plant growth. However, a poorly mixed fertilizer or a nutrient solution with pH outside the required range may keep the plant from using the nutrients. Other factors, such as lighting, air flow, dissolved oxygen, air relative humidity and temperature, can also keep plants outside their comfort zones.
Water quality impacts plant nutrition
A nutrient solution is water plus fertilizers. Maintaining good water quality in the nutrient solution is essential to a successful hydroponics nutrition program. The only way to know if your water quality is suitable for hydroponics is to test your water source (Table 1 shows adequate levels for water used in hydroponic systems). Many laboratories offer irrigation suitability testing packages; make sure they include pH, electrical conductivity (EC), alkalinity, and individual elements (nitrogen, phosphorus, potassium, calcium, magnesium, iron, manganese, and sodium). The test report will be helpful for calculating nutrient solution recipes, identifying water treatment needs, and establishing nutrient solution management practices that fit your farm.
To keep crops in their “comfort zone”, you will need to monitor and manage pH, EC, dissolved oxygen, temperature, and water alkalinity. In the following sections we will explain how these water quality parameters impact nutrient uptake and how to manage them to keep your plants healthy.
Table 1. Optimum ranges for water quality parameters to determine suitability of water sources for hydroponic cropping systems
| Parameter | Optimum range |
|---|---|
| pH | 5.5-7 |
| EC (dS/m) | 0.2-0.8 |
| Alkalinity | 40-160 ppm CaCO3 equivalent |
| Dissolved oxygen | >6 ppm |
| Total suspended solids | <30 ppm |
| Problematic ions | |
| Sodium | <50 ppm |
| Chlorine | <70 ppm |
| Sulfates | <90 ppm |
| Boron | <0.5 ppm |
| Fluor | <1 ppm |
| Calcium | <150 ppm |
| Magnesium | <75 ppm |
| Iron | <1 ppm |
| Manganese | <1 ppm |
| 1 ppm is the same as 1 mg/L (1 mg of the element in 1 liter of water) | |
pH
Water pH is a measurement of the concentration of hydrogen ions in the water. A scale with values ranging from 1 to 14 is used to represent pH, in which 7 is neutral, values under 7 are acidic, and values over 7 are basic. The water pH affects nutrient solubility and availability. If the pH is outside the optimum range, plants may not be able to uptake all the nutrients in the nutrient solution, or they may be too available becoming toxic to the plants. This has economic implications as those nutrient inputs are essentially wasted. As a rule of thumb, the optimum pH for most hydroponic crops (with exceptions) is from 5.5 to 6.5. Please consult with your local extension specialist for specific crop requirements.
Electrical conductivity
Electrical conductivity (EC) measures how well the nutrient solution conducts electricity. This is an indicator of dissolved nutrients in water, which may naturally occur in the water source or be added when dissolving fertilizers. While EC is a proxy for total dissolved ions, it does not confirm that each nutrient is at the target concentration. EC can be ‘on target’ while individual nutrients are unbalanced if the water source contributes significant ions or if fertilizer selection is mismatched.
Fertilizers are salts, which are compounds that have an ionic bond, positively charged ions (cations) bond with negatively charged ions (anions), neutralizing each other. When a fertilizer is correctly mixed in water, the water molecules break the ionic bonds of the fertilizers and the salts dissolve into ions. Dissolving fertilizers will increase ion concentration, increasing the water’s ability to conduct electricity. Measuring EC will indicate if a solution is properly mixed and the nutrients are available to the plants and tell you if fertilizer injectors are working properly. Some fertilizer labels will tell you what the expected EC of a solution at a set concentration is.
The roots of plants are only capable of taking up nutrients that are in ionic form, , properly mixed fertilizers will be dissolved from being salts and become available as ions. Each plant species will have a specific EC tolerance, but it is unlikely that your solution will surpass those levels when using recommended amounts. Ideally, you want your water source to have an EC below 1 dS/m (or mS/cm, mmho/cm), which will give you plenty of room to add fertilizers without increasing the EC to problematic levels.
Dissolved oxygen
In traditional soil cultivation the plant roots inhabit pore spaces that are filled with air when there is no rain. Roots need oxygen to properly function to support plant growth and nutrient uptake. A dissolved oxygen level over 6 ppm is optimum for hydroponics production. Low oxygen levels may inhibit growth and increase ethylene production.
The water temperature caps how much oxygen the water can hold. Warmer temperatures decrease the amount of oxygen that the water can hold. For example, at 35°C (95°F) with 5 dS/m electrical conductivity the oxygen solubility of water is 6.85 ppm, no matter how much air you pump in the water this value will not increase at that temperature. You can install water chillers to cool the water and increase the amount of oxygen water can hold. Oxygen availability is one example of how the nutrient solution temperature can affect plant health and nutrient uptake. Growers using aquaponic or deep-water culture (DWC) hydroponic systems, in which the roots are submerged in water, will need to monitor dissolved oxygen and water temperature.
Alkalinity
Alkalinity is a measure of the water’s capacity to neutralize acids. It is affected by the presence of bicarbonates and carbonates (calcium, magnesium, and sodium) and ions (hydroxides, phosphates, silicates, sulfides, and borates). Alkalinity, which measures the equivalents of calcium carbonate (CaCO3 ppm), does not correlate with pH and cannot be measured directly with a pH meter, meaning, high pH does not necessarily mean high alkalinity, and vice versa. Water alkalinity is measured through titration.
Water alkalinity impacts how often the water pH needs to be adjusted and how easy it is to adjust the pH. At high alkalinity (>160 ppm), higher amounts of acid are needed to change the pH. At levels of low alkalinity (<40 ppm), water pH changes constantly would require constant monitoring and adjustment.
Managing nutrient solutions
Nutrient solution management involves properly making nutrient solutions, monitoring key water quality parameters, and making the necessary adjustments to promote plant health. These are the steps farmers can follow to manage their nutrient solution:
- Test your water source
- Research nutrient requirements for your crops (nutrient levels and pH)
- Calculate how much fertilizer you need for the nutrient solutions
- Prepare nutrient solutions
- Measure pH and EC
- Adjust the pH as needed
- Constantly measure and adjust the pH and EC of the nutrient solution
Steps 1-3 are usually outsourced to testing laboratories and extension specialists. In Missouri, you can get your water tested at the MU Extension Soil and Plant Testing laboratory. You can share the water test report with your local extension specialist, and they will help you research nutrient requirements for your crops and create fertilizer recipes that match your farm’s water quality. Alternatively, you can download the calculator spreadsheet that accompanies this publication to make nutrient solution recipes. Farmers are then responsible for implementing steps 4-7, with available automation options for steps 5-7. This guide will show how to implement these steps.
Preparing nutrient solutions
Fertilizers must be properly added to nutrient solutions. The amounts and types of fertilizers required will change because crops require different nutrient levels at different growth stages.
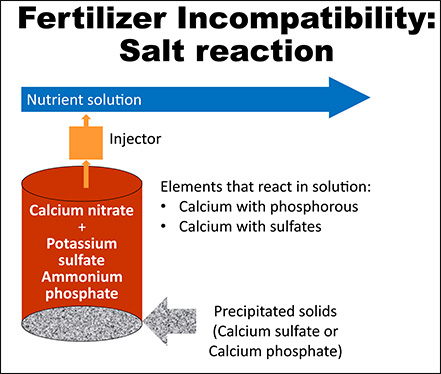
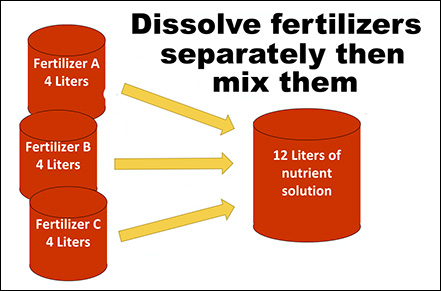
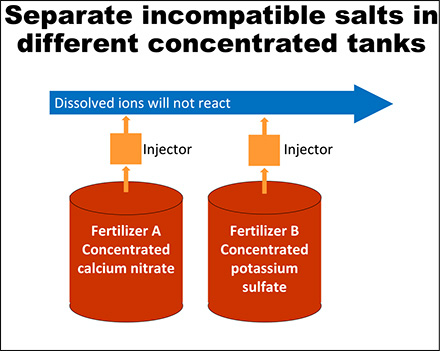
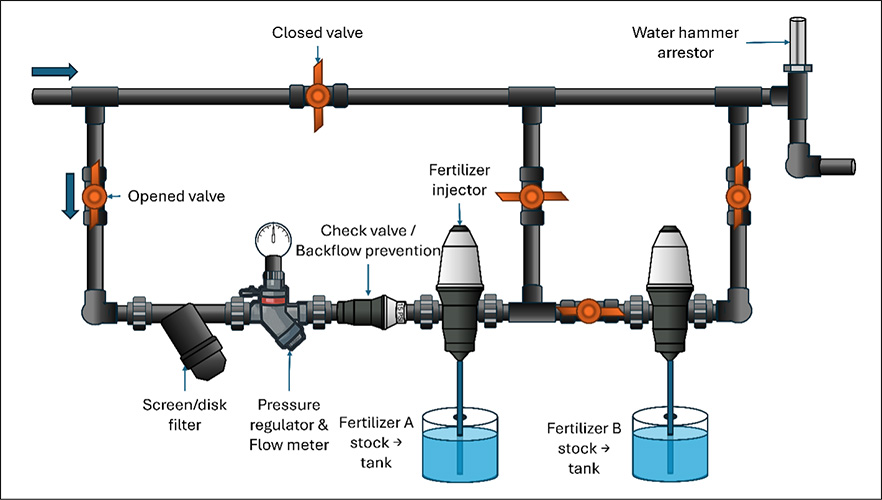
Hydroponic nutrient solution recipes may require at least two different fertilizer formulations. It is common for one of those formulations to provide calcium (15-0- 0), another formulation provides the rest of the nutrients (5-12-26), and in some cases, a third formulation may be needed. Properly mixing and dissolving the fertilizer formulations is crucial, because some fertilizers can react and precipitate out of solution when mixed together at the same time. Calcium from one formulation will immediately react with phosphorus and sulfate from the other formulation, creating insoluble particles that will sink to the bottom of the tank making them unavailable for plant roots (Figure 1). However, dissolving each fertilizer formulation separately will prevent this reaction from happening. Growers have the option to make the nutrient solutions by hand or using fertilizer injectors. Figure 2 shows how to mix fertilizers by hand and Figure 3 shows how to set up fertilizer injectors to prevent them from reacting. Also, a diagram with the recommended set up and fertilizer injection system components is depicted in Figure 4.
It is always important to check the fertilizer label to determine if the fertilizer is compatible with your hydroponic production system. These are key parts to check on the fertilizer label:
- Verify that the fertilizer is water-soluble: some formulations may have limited water solubility.
- Determine if the fertilizer is packaged in a single or multiple bag: some fertilizer formulations are usually sold as bundles marketed as “Part A and Part B” fertilizers. Fertilizers marketed as a single bag formulation will not provide essential nutrients at the optimum levels.
- Determine if the fertilizer has a dye: some formulations will come with a dye that will change the color of the water to indicate that a fertilizer has been mixed.
- Review the listing and concentration of all elements provided: this will help when making calculations needed to create nutrient solution recipes. Provide this information to your extension specialist.
- Review the expected EC: some fertilizer labels will indicate the expected EC after dissolving the fertilizer in water. This is helpful to determine if the fertilizers were properly mixed or when diagnosing problems with fertilizer injectors.
- Determine the potential acidity or basicity of a fertilizer: formulations with potential acidity help neutralize some of the water alkalinity when it is above 160 ppm CaCO3 equivalent. Formulations with potential basicity can add alkalinity to water sources under 40 ppm CaCO3 equivalent.
Monitoring and adjusting nutrient solutions
Nutrient solutions must be constantly measured and adjusted to meet crop requirements. The pH and EC of nutrient solutions change as dissolved fertilizers are added and as plants and microbes use water and nutrients over time. You must make the necessary adjustments to keep plants in their comfort zone.
Selecting meters
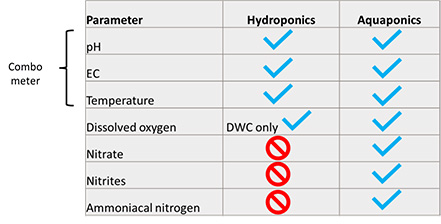
The type of meters needed to monitor nutrient solutions will depend on the type of hydroponic cropping system (Figure 5). The measurements are only as reliable as the meter itself; it is worthwhile to invest in good quality meters. Also, make sure you can calibrate the meters to ensure they provide consistently accurate measurements. For pH measurements, avoid using pH test strips in nutrient solutions because dyes in fertilizers can impact color.
Quality meters usually cost $100 to $300 as of 2025 (dissolved oxygen meters tend to cost more than $500). Select a waterproof and shockproof pH meter with replaceable probes. Select a portable meter that is easy to calibrate and store. A combination pH, temperature and EC meter, though often a bit more expensive, allows more monitoring options.
Meter calibration and care
The meter must also be periodically calibrated to maintain accurate measurements.
- Calibrate the meter once a week at two points: pH 4 and pH 7
- Avoid touching, scratching, or rubbing the pH probe glass bulb
- Store the pH probe in the storage solution or the pH 4 calibrating solution (NEVER STORE IT SUBMERGED IN WATER)
- Rinse with distilled or deionized water before every use, after calibration, in between samples, and before storing
- The pH probe will last 1 to 2 years; replace it when it can no longer be calibrated • An EC probe (combination pH EC meter) will last 2 to 5 years
- Use cleaning solutions provided by the manufacturer if a meter takes too long to take readings or calibrate.
Increasing pH
- To increase pH, a base must be added to the nutrient solution. These are common bases added to nutrient solutions:
- Potassium bicarbonate or potassium hydroxide (they provide potassium nutrient)
- Avoid using sodium bases because sodium can be toxic to plants
- Fertilizers with a high nitrate concentration (less than 25% of the total nitrogen from ammonia/urea)
- Avoid using calcium carbonate (lime) to lower pH because it has low solubility in water and can increase wear of the injector components.
Lowering pH
To decrease pH, add mineral and organic acids to the nutrient solution. These include, from the most expensive to least expensive: citric/acetic acid, phosphoric acid, nitric acid and sulfuric acid. Of these, citric/acetic is considered safer to handle than phosphoric and sulfuric acids, and nitric acid is considered the most dangerous for handling. Some acids provide additional plant nutrients.
The amount of acid needed to change the pH will depend on the alkalinity of the water. The higher the alkalinity, the more acid you will need to make a change in pH. There is an online calculator that will help you determine how much acid to add to your water source. You will need to input the results of your water test report to use the calculator.
There are injector systems in the market that can automatically monitor and adjust the pH and EC of the water.
Monitoring rootzone pH and EC in hydroponic systems based on soilless media
So far, we have talked about managing nutrient solutions to ensure optimal nutrient availability. The recommendations are suitable for water-based hydroponic systems (roots in direct contact with the nutrient solutions). However, in soilless media, the roots are in direct contact with growing media which can impact nutrient availability.
You can safely assume that the pH and EC in the rootzone will be similar to the pH and EC of the nutrient solution when using inert growing media (perlite, expanded clay pellets, gravel, highly lignified coconut fiber, and rockwool). When using organic-based media (wood fiber, peat moss, etc.), the components of the nutrient solution will react with the components and microorganisms in the organic matter, resulting in a rootzone pH and EC that is different from that of the nutrient solution. You can implement the Pour-Thru or the Saturated Media Extract (SME) methods to measure the rootzone pH and EC in organic growing media.
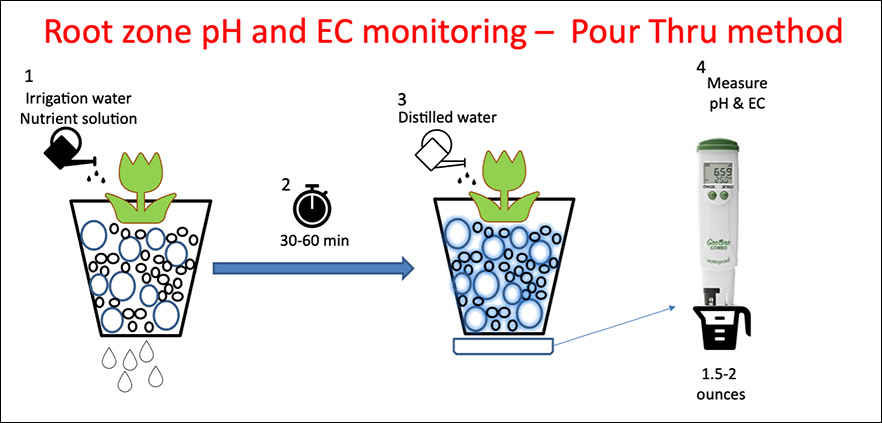
Pour-Thru method is non-destructive and is not suitable for crops that are sub-irrigated (watered from the bottom).
Steps (See Figure 6)
- Irrigate the crop with the nutrient solution
- Stop watering the crop when you see water draining from the pots
- Let sit for 30-60 minutes
- Place the container on a saucer
- Slowly pour distilled water until you collect 1.5 to 2 ounces of drained solution in the saucer
- Place the drained solution in a cup and measure the pH and EC in that solution.
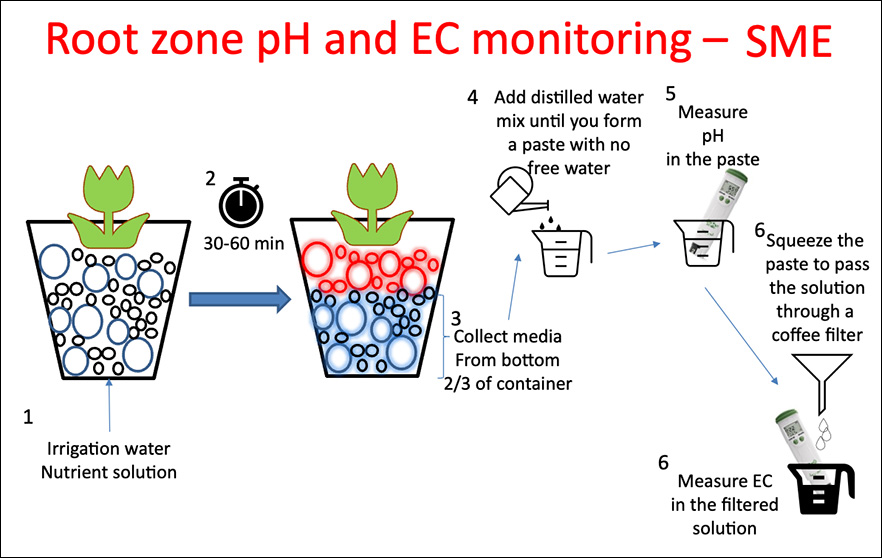
Saturated Media Extract (SME) is a destructive method, suitable for both sub- and over-irrigated crops, not suitable when using slow-release fertilizers.
Steps (See Figure 7)
- Irrigate the crop with the nutrient solution
- Stop watering the crop when you see water draining from the pots.
- Let sit for 30-60 minutes
- Collect media from the bottom 2/3 section of the container
- Place the collected media in a cup
- Slowly add distilled water and mix until forming a paste with no free-flowing water
- Place the meter directly in the paste and measure the pH 8. Squeeze liquid from the paste into a coffee filter in a funnel and collect the filtered liquid 9. Measure the EC in the filtered solution
Summary
Managing nutrient solutions is central for successful hydroponic systems. In review, these are the main steps in managing nutrient solutions:
- Test your water source
- Research nutrient requirements for your crops (nutrient levels and pH)
- Calculate how much fertilizer you need for the nutrient solutions
- Prepare nutrient solutions
- Measure pH and EC
- Adjust the pH as needed
- Constantly measure and adjust the pH and EC of the nutrient solution.