Nitrogen is important to all life. Nitrogen in the atmosphere or in the soil can go through many complex chemical and biological changes, be combined into living and non-living material, and return back to the soil or air in a continuing cycle. This is called the nitrogen cycle.
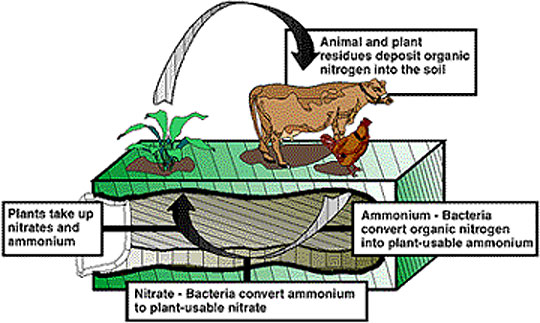
Figure 1
Oversimplifying, the nitrogen cycle works this way.
A basic look at the nitrogen cycle
Plants need nitrogen to grow, develop and produce seed. The main source of nitrogen in soils is from organic matter. Soils in Missouri commonly contain one to four percent organic matter. Organic matter largely arises from plant and animal residues. The nitrogen in organic matter is largely in organic forms that plants cannot use. Bacteria found in soils convert organic forms of nitrogen to inorganic forms that the plant can use. Nitrogen is taken up by plant roots and combined into organic substances in the plant, such as enzymes, proteins and chlorophyll. Chlorophyll gives the plant its green color. When the plant dies, it decays and becomes part of the organic matter pool in the soil. The basic nitrogen cycle is illustrated in Figure 1. It shows nitrogen changing from organic matter in the soil, to bacteria, to plants and back to organic matter.
- Plant and animal wastes decompose, adding nitrogen to the soil.
- Bacteria in the soil convert those forms of nitrogen into forms plants can use.
- Plants use the nitrogen in the soil to grow.
- People and animals eat the plants; then animal and plant residues return nitrogen to the soil again, completing the cycle.
Another way nitrogen enters the cycle is as inorganic nitrogen from the atmosphere and factories. The concern with these forms is that the incremental amount of nitrates they add to the nitrogen cycle may threaten groundwater.
- Rain storms contribute atmospheric nitrogen through rain drops that reach the soil.
- Legumes, such as soybeans, alfalfa and clovers, are plants that can convert atmospheric nitrogen into plant-usable nitrogen.
- Factories that produce nitrogen fertilizers add nitrogen to the soil when farmers and gardeners "feed" their crops.
- Nitrogen in sewage sludge from municipal waste plants can be used to fertilize farm fields.
Ways nitrogen is lost to the cycle
For the most part, the nitrogen cycle is soil based. Nitrogen is lost from the cycle in four ways:
- Denitrification
Bacteria change nitrate in the soil to atmospheric nitrogen, which joins the atmosphere. - Volatilization
Turns urea fertilizers and manures on the soil surface into gases that also join the atmosphere.
Together, these first two processes account for most of the nitrogen lost to the cycle — a concern for soil fertility.
- Runoff
Carries the nitrogen in fertilizers and manure and the nitrogen in the soil into our rivers and streams — a concern for water quality. - Leaching
Carries nitrates so deep into the soil that plants can no longer use them, producing a dual concern — for lost fertility and for water quality, as nitrates enter the groundwater and the wells that provide our drinking water.
More about the nitrogen cycle
The largest single source of nitrogen is the atmosphere. It is made up of 78 percent of this colorless, odorless, nontoxic gas. However, plants are unable to use nitrogen as it exists in the atmosphere. Nitrogen from the air (N2) enters the nitrogen cycle through several unique types of microorganisms that can convert N2 gas to inorganic forms usable by plants. Some of these microorganisms live in the soil, while others live in nodules of roots of certain plants.
Nitrogen also can enter the cycle from other sources besides the air, manure and decaying plant materials. Nitrogen also can enter the cycle from the application of commercial nitrogen fertilizers.
Nitrogen can be lost from the cycle. It can be lost to the atmosphere, removed by harvesting crops or lost to surface water or groundwater. However it is lost, nitrogen can enter the cycle again through one of the processes discussed above or through other processes. These additional pathways of gains and losses to the nitrogen cycle are illustrated in Figure 2.
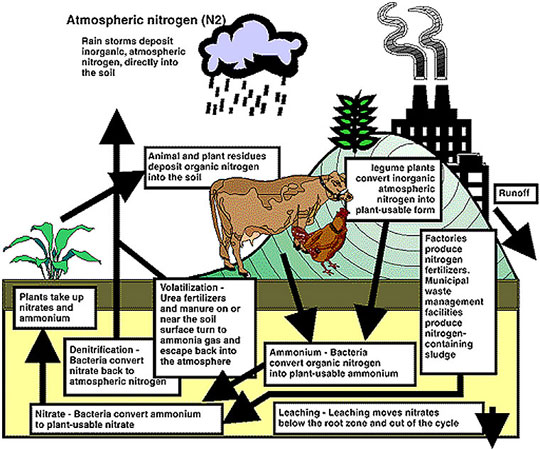
Figure 2
A more comprehensive look at the nitrogen cycle.
Impact on water quality
Nitrogen becomes a concern to water quality when nitrogen in the soil is converted to the nitrate (NO3-) form. It is a concern because nitrate is very mobile and easily moves with water in the soil. The concern of nitrates and water quality is generally directed at groundwater. However, nitrates can also enter surface waters such as ponds, streams and rivers. The presence of nitrates in the soil are largely the result of natural biological processes associated with the decomposition of plant residues and organic matter. Nitrates can also come from rainfall, animal manure and nitrogen fertilizers.
Whether or not nitrates actually enter groundwater depends on underlying soil and/or bedrock conditions, as well as the depth to groundwater. If depth to groundwater is shallow and the underlying soil is sandy, the potential for nitrates to enter groundwater is relatively high. However, if depth to groundwater is deep and the underlying soil is heavy clay, groundwater contamination from nitrates is not likely.
Once nitrates get into the groundwater, the greatest concerns are for infants less than one year old and for young or pregnant animals. High levels of nitrates can be toxic to newborns, causing anoxia, or internal suffocation. Seek alternative water sources if nitrate levels exceed the health standard of 10 ppm nitrate-N. Do not boil water to eliminate nitrates. It increases nitrate levels rather than decreasing them. The most common symptom of nitrate poisoning in babies is a bluish color to the skin, particularly around the baby's eyes and mouth. These symptoms of nitrate toxicity are commonly referred to as the "blue-baby" syndrome.
The initial draft of this publication was written by Karen DeFelice, former associate extension agronomist; Nyle Wollenhaupt, former state extension agronomist; and Daryl Buchholz, state extension agronomist. This material is based upon work supported by the United States Department of Agriculture, Extension Service, under special project number 89-EWQI-1-9203.